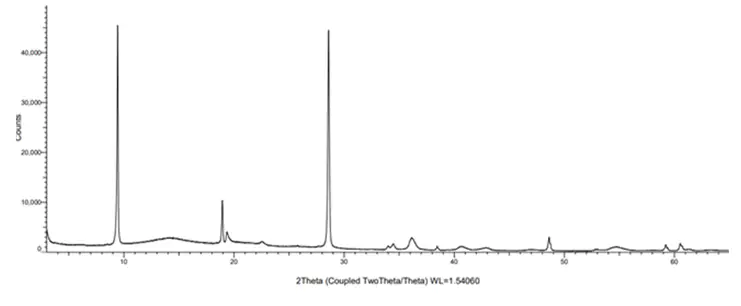
The introduction of USP <901> in December 2025 has significantly raised expectations for the detection of asbestos in pharmaceutical talc.
For manufacturers and suppliers, this isn’t just a regulatory update, it’s a shift that directly impacts product safety, compliance risk and time to market.
Talc is widely used across pharmaceuticals, cosmetics and personal care products, yet its natural proximity to asbestos deposits means contamination remains a genuine and ongoing risk. With asbestos linked to serious health conditions including asbestosis, mesothelioma and lung cancer, regulators are placing greater emphasis on trace level detection and robust analytical evidence.