Types of autoclaves
There are different types of autoclaves, based on their operating principles. The most common to pharmaceuticals and healthcare facilities are:
- Gravity displacement autoclaves: Steam enters from top/sides and pushes air downward out of the chamber through a drain. Suitable for liquids, simple instruments, non porous loads. These are sometimes referred to as Class N autoclaves. The ‘N’ stands for naked solid products.
- Pre vacuum (high speed) autoclaves: A vacuum pump is used to remove air before steam entry, improving steam penetration into porous loads, wrapped items or hollow instruments. These are sometimes referred to as Class B autoclaves - these are the highest standard. The ‘B’ represents ‘big’.
- Autoclaves that combine gravity air displacement methods with limited vacuum systems. These are mid-way between N and B class and are sometimes referred to as Class S (with the S representing ‘special’). While Class S autoclaves offer greater versatility than Class N models, they do not provide the same level of effectiveness for complex materials as Class B autoclaves.
These autoclaves differ primarily in their sterilisation capabilities and air removal methods. The classes are defined by the European Standard EN 13060: 2025 2 (‘Small steam sterilizers’). In summary:
| Class |
Method |
Load Type |
Best Use Case |
| B |
Fractionated vacuum |
All (wrapped, porous, hollow) |
Hospitals, complex dental tools |
| S |
Partial/no vacuum |
Specific (manufacturer defined) |
Basic wrapped, solid tools |
| N |
Gravity displacement |
Simple (unwrapped, solid) |
Basic, non-hollow tools |
The standard EN 285: 2014 3 applies to large steam sterilisers (≥ 54 litres or a full-sized sterilisation unit). The standard itself does not classify machines into types (like ‘Class B’), whereas it defines the performance, design and testing requirements for these large, usually pre-vacuum, autoclaves (in essence Class B).
How an autoclave cycle works: Step by step
While there are differences with autoclave design, there are some common factors which apply. These are set out below:
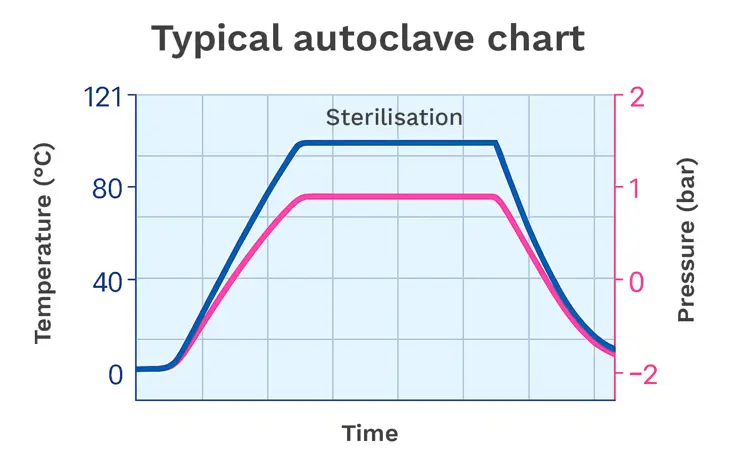
Figure 1: A typical autoclave chart showing temperature and pressure rising through conditioning, plateauing during sterilisation and dropping during exhaust (image designed by Tim Sandle)
(1) Loading
- Items must be cleaned and arranged to allow free steam circulation
- Wrapped or hollow instruments require careful orientation
- Use chemical and biological indicators where required
(2) Chamber sealing
- The door locks with pressure safety interlocks. These seal the autoclave chamber, meaning the door cannot open when the chamber is pressurised. A heat resistant gasket ensures a steam tight seal is in place
(3) Air removal (critical step)
- Air acts as an insulator, preventing steam from reaching sterilisation temperature - therefore, air must be removed
- The mode of air removal depends on the autoclave type. Gravity units push air out the drain, while pre vacuum units extract air with a vacuum pump
(4) Steam admission and heating
- Steam is introduced until the chamber reaches the programmed temperature and pressure (e.g., 121 °C or 134 °C)
- Steam condenses on cooler surfaces, releasing high energy directly onto microbial cells, causing protein denaturation and cell death
(5) Sterilisation hold (exposure phase)
Depending on load type and mass, common validated conditions for the hold stage are:
- 121 °C for 15-20 min
- 134 °C for 3-5 min
(6) Exhaust / depressurisation
- Once the hold time has elapsed, the autoclave needs to be rendered safe for users. This requires steam to be vented safely as the pressure drops to atmospheric level
(7) Drying
- The drying stage is especially important for wrapped sets to avoid ‘wet loads’ (which are a sign of non-sterility). This arises because moisture insulates the load, prevents achieving temperature and creates an environment for microbial growth. Wet load issues are also caused by poor loading configurations
Key performance and technical requirements
Under EN 285, the important performance and technical requirements are:
- Steriliser applicability: This defines what the autoclave is to be used for and what is the appropriate type
- Scope: This covers mechanical construction, piping, control systems, safety and performance, but generally excludes sterilisers for flammable substances or bio-waste
- Steam quality tests (see Figure 2): This sets out the requirements for steam quality, including:
- Non-condensable gases: Limit ≤ 3.5% v/v. Non‑condensable gases are gases present within steam that do not liquefy (condense) under the temperature and pressure conditions used in steam sterilisation. Instead of turning into water when steam condenses, they remain as gas pockets such as air, carbon dioxide, nitrogen, hydrogen or other trace gases. Excessive levels can lead to non-sterility (through block steam penetration, reduced heat transfer, the creation of cold spots), even where biological indicators pass
- Dryness value: Limit ≥ 0.95. With steam dryness, steam must be saturated and collapse on contact to transfer heat. Excess wetness causes wet loads, poor dryness fraction (<97%) prevents lethality
- Superheat: Limit ≤25.0°C. Superheat is the temperature of steam above its saturation point. Once steam has reached the temperature at which it should condense at a given pressure (its saturation temperature), any additional temperature above is called ‘superheat’. Too much superheat means the steam is too dry, which reduces heat transfer efficiency. Steam must condense on the load to release latent heat (the lethal step)

Figure 2: Steam quality (designed by Tim Sandle)
Validation and testing
The validation and testing of autoclaves require specific, rigorous tests to be conducted to ensure sterilisation efficacy, including:
- Thermometric tests: These range from small and full load tests on standard towel packs
- Air detector tests: This ensures proper air removal
- Air leakage tests: Limit ≤ 10-11 mbar/min
- Equilibration time: For chambers up to 800 litres, it must not exceed 15 seconds - for larger chambers, it must not exceed 30 seconds
Effective sterilisation must be verified using chemical (ISO 11140-1:2014) 4 and biological indicators (general requirements: ISO 1138-1: 2017 5, most heat: ISO 11138-3:2017) 6. Of these different indicators, chemical indicators verify exposure to sterilisation conditions and biological indicators (e.g., Geobacillus stearothermophilus) confirm lethality.




