
Although not used as an ingredient, dioxane (1,4-dioxane; CAS 123-91-1) may be present at trace levels in cosmetics, household cleaning and personal care products, being an unintentional by-product of the manufacturing process of ethoxylated ingredients such as detergents, emulsifiers, foaming agents and solvents.
With many personal care and cleaning products being washed down the drain, 1,4-dioxane is released into wastewater. Listed in group 2B as a possible human carcinogen by the International Agency for Research on Cancer (IARC), dioxane is highly mobile, persistent in water and not easily removed by wastewater and drinking water treatments. Due to these properties, this compound is a contaminant of emerging concern, and several markets are looking to adopt stricter regulatory measures regarding its presence in these products.
In 2020, the United States Environmental Protection Agency (EPA) released a risk evaluation for dioxane, which showed no unreasonable risks to the environment, consumers or the general population. However, in 2021, the EPA re-opened this evaluation to include additional exposure pathways (e.g., drinking water, ambient air) and conditions of use (e.g., industry by-product), which were previously excluded. A draft supplement to the 1,4-dioxane testing evaluation is expected to be released in June 2023 for public comment.
Currently, the United States Food and Drug Administration (FDA) has not established a federal limit for the permissible amount of dioxane in cosmetics. Although it hasn’t conducted an independent risk assessment, the FDA has been periodically monitoring dioxane levels in cosmetic products sold in the United States since the late 1970s. Following the recommendations to implement changes in the manufacturing processes to reduce this contaminant, the FDA has reported a significant decrease of dioxane levels in cosmetics over the years.
Due to concerns regarding the general population’s exposure to dioxane through drinking water, the state of New York has recently adopted a law to establish, for the first time in the country, the permissible limits of dioxane in cosmetics, household cleaning and personal care products offered for sale in the state (New York Senate Bill No. S4389B). This law limits the dioxane levels to 10 parts per million (ppm) in cosmetics and to 2 ppm in household cleaning and personal care products from 31st December 2022. This will be further reduced to 1 ppm by 31st December 2023.
Manufacturers may apply for a one-year waiver for a specific product based on the submission of proof that measures to reduce the presence of dioxane have been taken. Upon the waiver expiration, any product exceeding the dioxane levels established in the New York Bill cannot be sold in the state. Non-compliances will be subject to penalties up to $1000/day per violation and $2500/day for second violations. From 2025, the imposed limits for dioxane will be reviewed by the New York Department of Environmental Conservation every two years and it will be considered whether to lower them.
Although not restricting its use, California has already adopted warning or disclosure requirements for dioxane in consumer products offered for sale in the state. Dioxane has been listed on Proposition 65 (Safe Drinking Water and Toxic Enforcement Act of 1986) since 1998 due to its classification as a potential human carcinogenic agent. A No Significant Risk Level (NSRL) of 30 µg/day was established. If the estimated level of exposure to dioxane exceeds this safe harbor level, the manufacturer is required to provide a clear warning to the consumer. Under the 2005 California Safe Cosmetics Act, manufacturers of cosmetics sold in the state are required to provide a list of products containing dioxane. In 2013, dioxane was identified as a “candidate chemical” that may warrant further research by the California Department of Toxic Substances Control. Since 2020, all manufacturers of cleaning products are required to disclose on their website, as well as the product label if dioxane is present at levels above 10 ppm (Cleaning Product Right to Know Act of 2017). Following New York state action, California has launched an initiative that may result in additional regulatory limits on the use of dioxane in cosmetics, household cleaning and personal care products.
The European Union (EU) has not formally set limits for the amount of dioxane in cleaning products. Although its use as an ingredient is banned in the European Union (EU) for cosmetics and personal care products, dioxane may still be present as a contaminant at trace levels. The European Cosmetics Regulation EC 1223/2009 allows the unintentional presence of trace levels of contaminants or impurities when this is technically unavoidable, providing that there is compliance with good manufacturing practice and no risk to consumers’ health. The European Commission Scientific Committee on Consumer Safety (SCCS) have concluded that trace levels up to 10 ppm of dioxane in cosmetic products is considered safe for consumers.
In 2021, the European Chemicals Agency (ECHA) added dioxane to its list of substances of very high concern due to its carcinogenic and intrinsic chemical properties, which demonstrated scientific evidence of probable serious effects to the environment and human health. Similarly, in the United Kingdom, the Health and Safety Executive (HSE) and the Environment Agency added dioxane to the list of substances that require a regulatory management options analysis. This may lead to stricter regulatory measures on the presence of dioxane in cosmetics, household cleaning and personal care products across Europe.
In 2019, the Association of South East Asian Nations (ASEAN) updated its Guidelines on Limits of Contaminants for Cosmetics, setting new limits for the amount of dioxane permissible in cosmetic products in ten member states. This update came into force in June 2020 and limits the dioxane levels to 25 ppm in cosmetics. This will be further reduced to 10 ppm by 19th June 2023.
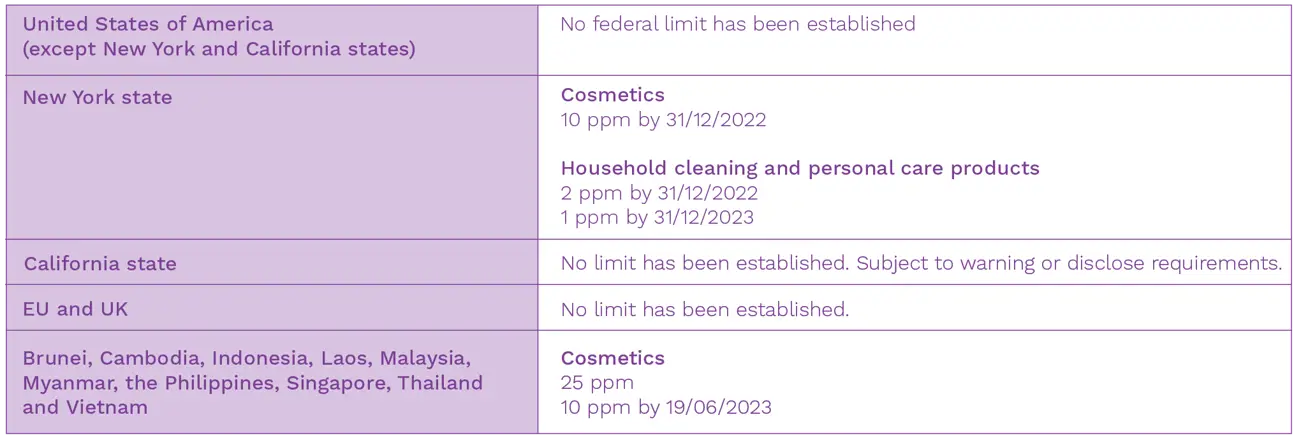
Table 1: Permissible limits of dioxane as a contaminant in cosmetics, household cleaning and personal care products.
As other markets start to look at dioxane levels, it is recommended that manufacturers start to evaluate affected products and consider the potential need for product reformulation in order to take into account any new established limits. RSSL has extensive technical expertise and experience in contaminant identification and investigations, we can support with 1,4-Dioxane analysis in cosmetics, household cleaning and personal care products. Our experts can investigate the level of dioxane in your products, support with reformulation in anticipation of stricter limits, develop bespoke testing methods and provide expert guidance to ensure you are meeting the regulatory requirements. Click below to find out more about 1,4-Dioxane testing or contact us via the form below.